The Cholinergic Anti-Inflammatory Pathway — The Vagus Nerve as the Body's Master Switch
The Cholinergic Anti-Inflammatory Pathway — The Vagus Nerve as the Body's Master Switch
Thesis
Your body already has a neural circuit that shuts down dangerous inflammation in real time — the vagus nerve. When activated, it releases acetylcholine directly at immune cells, suppressing the cytokine storm before it damages tissue. This isn't metaphor. It's a hard-wired reflex, as real as the knee-jerk reflex, and it's been hiding in plain sight since 2002.
Key Questions
- How does a nerve directly control immune function?
- What is the mechanism that converts neural signals into immune suppression?
- Why does cutting the vagus nerve make inflammation dramatically worse?
- Can we therapeutically activate this pathway to treat chronic inflammatory diseases?
Supporting Research
Tracey, K.J. (2002). The inflammatory reflex. Nature, 420, 853–859.
DOI: 10.1038/nature01321
PubMed | Nature
Borovikova, L.V. et al. (2000). Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature, 405, 458–462.
The Inflammatory Reflex: A Neural Circuit for Immune Control
Inflammation is essential for survival — it fights infection and heals wounds. But uncontrolled inflammation is a killer. Sepsis, rheumatoid arthritis, Crohn's disease, and post-surgical complications all share one feature: the inflammatory response has gone haywire. Kevin Tracey's 2002 Nature paper revealed something remarkable: the nervous system has a dedicated circuit to prevent exactly this.
The reflex has two arms:
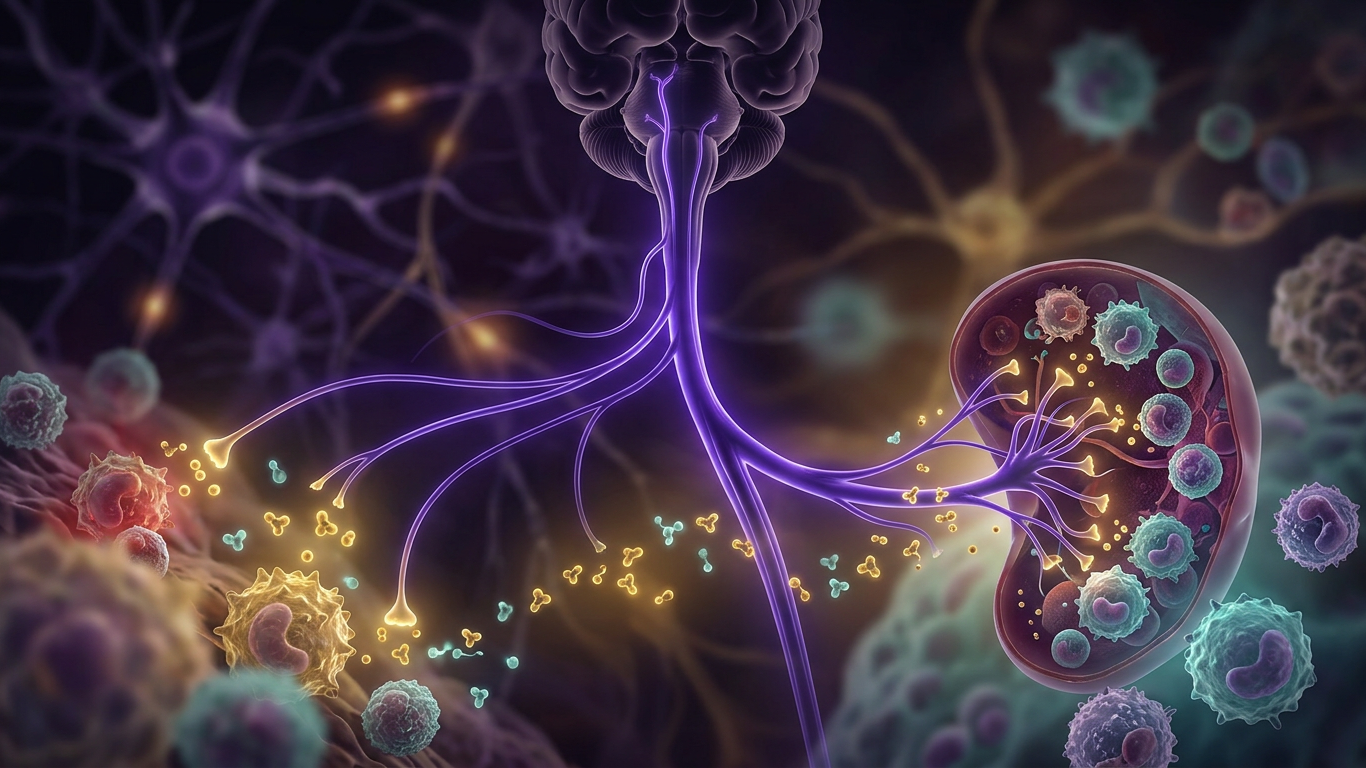
- Afferent (sensory) arm: Pro-inflammatory cytokines (TNF, IL-1, HMGB1) released from damaged or infected tissues activate vagus nerve sensory fibers. These signals travel to the nucleus tractus solitarius (NTS) in the brainstem, then to the hypothalamus.
- Efferent (motor) arm: The brain triggers anti-inflammatory output via the vagus nerve, which innervates the spleen, liver, heart, lungs, gut, and kidneys.
This creates a real-time feedback loop: inflammation is detected → the vagus nerve fires → inflammation is suppressed. The system prevents local inflammation from spilling over into a systemic crisis.
The Cholinergic Mechanism
The efferent arm works through a specific molecular pathway:
- Vagus nerve terminals release acetylcholine (ACh) near tissue macrophages
- ACh binds to α7 nicotinic acetylcholine receptors (α7nAChR) on macrophages
- This inhibits synthesis of pro-inflammatory cytokines (TNF, IL-1, IL-18, HMGB1) at a post-transcriptional level
- Anti-inflammatory cytokines like IL-10 are unaffected — the pathway is selective
Nicotine mimics acetylcholine, confirming the nicotinic (not muscarinic) mechanism. The pathway specifically targets tissue-resident immune cells, not circulating monocytes.
Experimental Proof
Three types of experiments confirmed this:
- Vagotomy (cutting the vagus): TNF levels spike, endotoxin sensitivity increases, inflammation models worsen dramatically
- Electrical VNS: Suppresses TNF in spleen, liver, and heart; reduces serum cytokines during sepsis; protects against shock; attenuates arthritis
- Pharmacological: CNS-acting drugs like CNI-1493 only work when the vagus nerve is intact — remove the nerve, the drug stops working
Why This Matters
This discovery launched the field of bioelectronic medicine. Vagus nerve stimulation devices were already FDA-approved for epilepsy — now they were being tested for rheumatoid arthritis, Crohn's disease, and sepsis. The implications extend beyond medicine: vagal tone (measured by heart rate variability) may be a biomarker for chronic inflammation risk. Activities that boost vagal tone — breathing exercises, meditation, cold exposure — could theoretically enhance this anti-inflammatory reflex.
Experimental Predictions
- Higher resting vagal tone should correlate with lower baseline inflammatory markers (CRP, IL-6, TNF)
- Chronic VNS should reduce disease activity in inflammatory conditions
- Pharmacological enhancement of cholinergic signaling (AChE inhibitors) should reduce systemic inflammation
- Vagal tone should decline with age, paralleling increased inflammatory disease incidence
Have a question?
Have a question about something specific? Send us a message.
Visit VagusSkool.com/contact — we'll try to get back to you within 24 hours.